In December 2008, the U.S. Food and Drug Administration (FDA) approved Rebiana, also known as Reb A or rebaudioside A, found in the stevia leaf. Stevia is derived from the leaves of a South American shrub and has been used to sweeten foods in Brazil, Paraguay, Japan, South Korea and China for many centuries. Reb A is one of two compounds; the other being stevioside, found in the stevia leaf. Reb A is less bitter and sweeter than stevioside. Rebiana is approved by the FDA as “generally recognized as safe” (GRAS).

Today, Truvia and PureVia are available in packets similar to other sugar substitutes such as Splenda, Equal and Sweet n’ Low, and will soon be seen in a growing number of sugar-free products. The main ingredients are Reb A (from the stevia leaf) and Erythritol (a sugar alcohol with no effect on glucose levels and little or no laxative effect).
Truvia™ can be found at Whole Foods, Walmart and other grocery stores.
- A Coca-Cola and Cargill product
- Over 200 times sweeter than sugar
- Ingredients: Rebiana, Erythritol, natural flavors
- Rebiana or Rebaudioside is the sweet extract from the stevia leaf – for sweetness
- Erythritol is a sugar alcohol derived from fruits like grapes and melons to improve taste and reduce bitterness
- Natural flavors to enhance flavor
- Found in products such as: sweetener packets, Sprite Green and Odwalla juice drinks
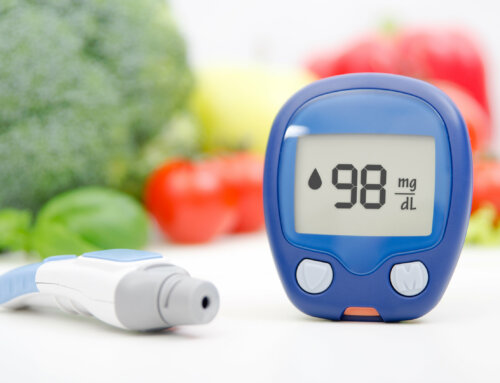
- Zero calories and no effect on glucose levels
PureVia™ can be found at grocery stores.
- PepsiCo and Whole Earth Sweetener co-product
- Over 200 times sweeter than sugar
- Ingredients: Rebiana, Erythritol, natural flavors
- Rebiana or Rebaudioside is the sweet extract from the stevia leaf to improve sweetness
- Erythritol is a sugar alcohol derived from fruits like grapes and melons to improve taste and reduce bitterness
- Isomaltulose (found in honey and sugar case juice)
- Cellulose powder is a dietary fiber to enhance texture
- Natural flavors to enhance flavor
- Found in products such as sweetener packets and SoBe Lifewater
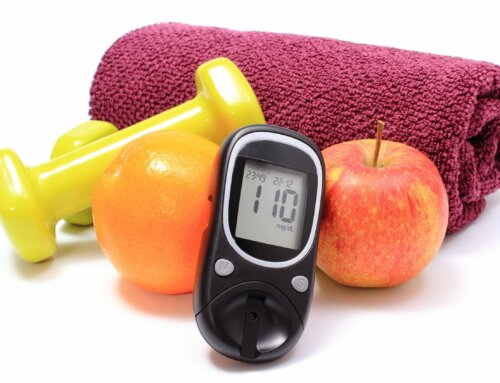
- Zero calories and no effect on glucose levels
Use moderately. Moderate use is 4 servings a day: 4 packets or food servings similar to food exchanges (8 ounce drink, etc.)




Leave A Comment